
235U decays via alpha decay (thorium-231) into 231Pa with a half-life of ~7×10 8 years. 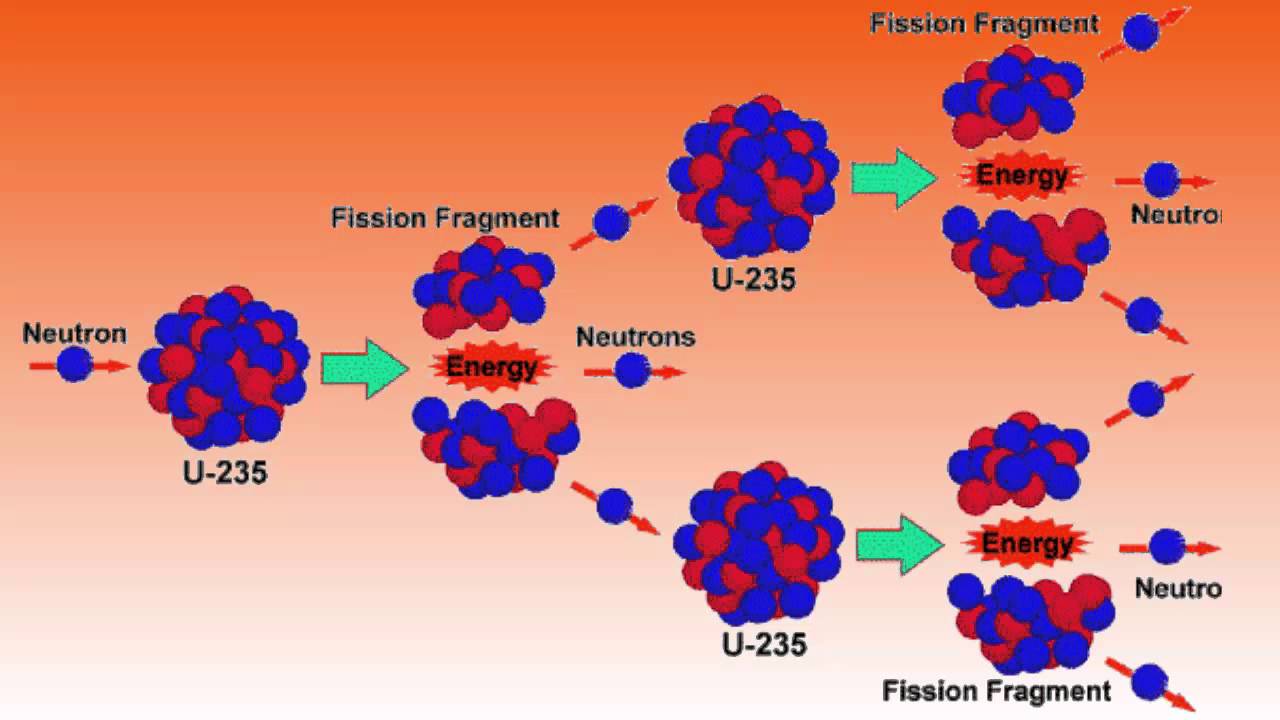
235U is the only existing fissile nucleus from naturally occurring isotopes, and therefore it is a highly strategic material. 235U belongs to the group of fissile isotopes. Its specific activity is very low ~3.4×10 -7 Ci/g. 238U occasionally decays by spontaneous fission with the probability of 0.000055%. 238U decays via alpha decay to 234Th with a half-life of ~4.5×10 9 years. 238U belongs to the group of fertile isotopes. The main isotopes, which have to be considered in the fuel cycle of all commercial light water reactors, are: These are produced by transmutation in nuclear reactors from 235U and 232Th. In nuclear reactors we have to consider three artificial isotopes, 236U, 233U and 232U. On the other hand, these isotopes (except 234U) belong to primordial nuclides because their half-life is comparable to the age of the Earth (~4.5×10 9 years for 238U). All three naturally occurring isotopes of uranium ( 238U, 235U, and 234U) are unstable. Differences in the half-lives cause the abundance of isotopes in nature. Natural uranium also consists of two other isotopes: 235U (0.71%) and 234U (0.0054%). Therefore the atomic mass of the uranium element is close to the atomic mass of the 238U isotope (238.03u). Natural uranium consists primarily of isotope 238U (99.28%). Significant concentrations of uranium occur in some substances such as uraninite (the most common uranium ore), phosphate rock deposits, and other minerals. Uranium also occurs in seawater and can be recovered from ocean water. Uranium is commonly found at low levels (a few ppm – parts per million) in all rocks, soil, water, plants, and animals (including humans). He was the first to discover the process of radioactivity. 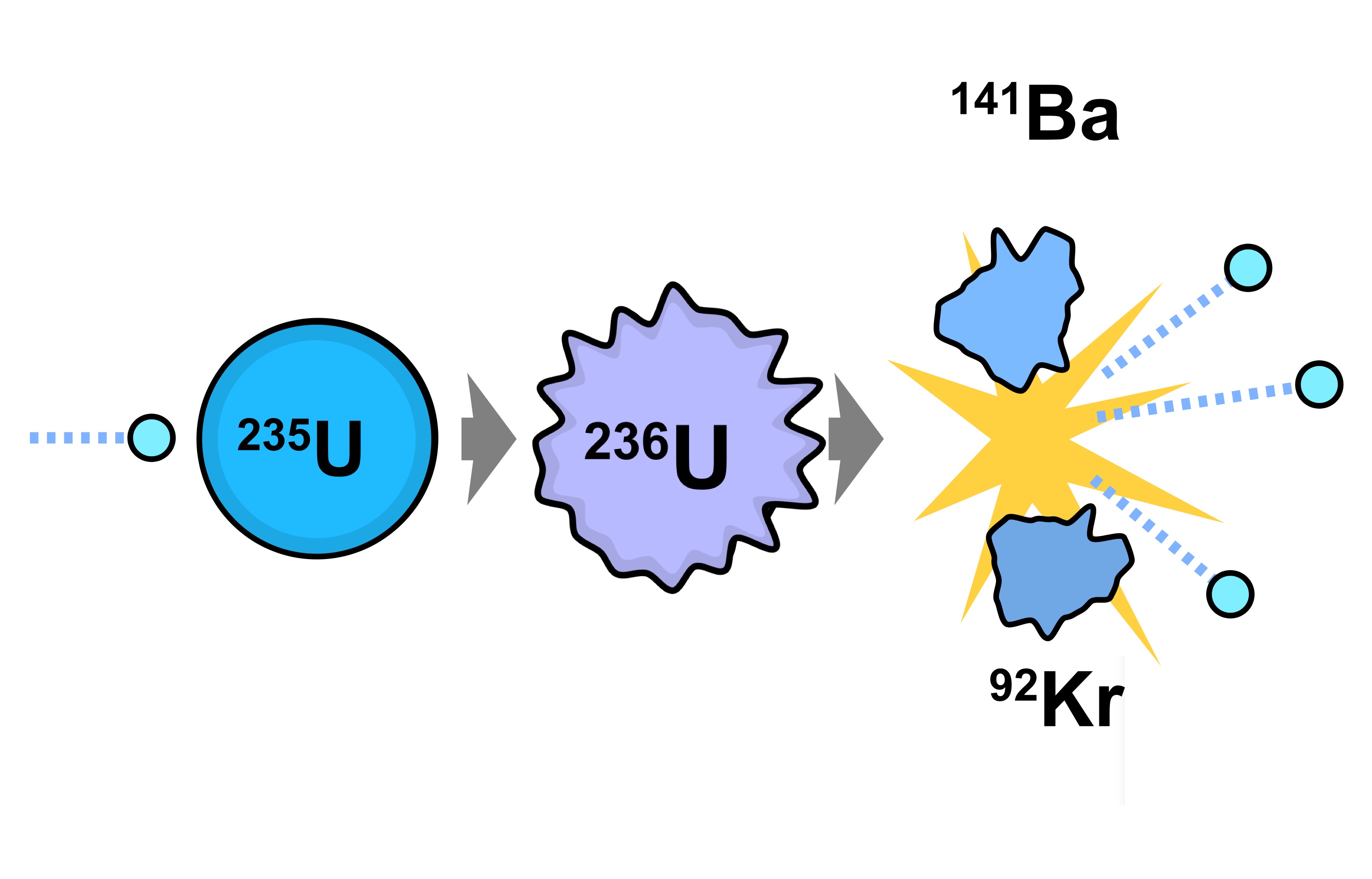
He discovered that uranium minerals could expose a photographic plate through another material. Henri Becquerel discovered uranium to be radioactive in 1896. It was first isolated as a metal in 1841 by Eugene-Melchior Peligot. He named the newly discovered element after the planet Uranus, which had been discovered eight years earlier. Uranium was discovered in 1789 by Martin Klaproth in the mineral called pitchblende (uraninite). Uranium is a naturally occurring chemical element with atomic number 92, which means there are 92 protons and 92 electrons in the atomic structure.
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
